Bone is not a static structure. It is a living mechanical system, where everyday loading drives fluid flow, activates osteocytes, and regulates remodeling. With age, this force-to-signal loop shifts. Osteocytes change their mechanophenotype, the microenvironment that transmits forces becomes more heterogeneous, and senescent cells can reshape local signaling and inflammation. The result is not only altered bone mass, but altered mechanosensory capacity and impaired repair.
In this Faces of Mechanobiology guest blog, Maryam Tilton Ph.D., Assistant Professor at The University of Texas at Austin and Director of the BioMATTER Lab, explores “skeletal mechano-aging” through the lens of osteocyte senescence. She shares how her lab pairs molecular and morphometric readouts with live single-cell biomechanical phenotyping using optical fiber interferometry nanoindentation on the Optics11 Life Pavone platform. From stress-induced senescence and its biophysical signatures to engineered microenvironments for repair, including a visible-light DLP 3D-printed TPMS hydrogel scaffold with localized oxygen release, this article connects mechanobiology across scales, from single cells to regeneration.
Mechanobiology of the Aging Skeleton: Osteocyte Senescence and Engineered Microenvironments for Repair
Bone is a living mechanical system. Every day, loading deforms mineralized tissue, drives fluid through the lacunar-canalicular network, and triggers osteocytes to coordinate bone remodeling and homeostasis. This is the core feedback loop of bone mechanobiology: osteocytes encode mechanical inputs into biochemical signals that reshape the pericellular and tissue microenvironment that transmits the next round of force.
Aging does not simply weaken this loop; it shifts its operating point, meaning the same load can yield a different cellular output. Osteocytes change their mechanophenotype, and the local microenvironment that transmits deformation and fluid flow to them also changes. With aging-related remodeling, the pericellular matrix and lacunar-canalicular transport can shift, and the tissue becomes more mechanically heterogeneous. In parallel, senescent cells, including senescent osteocytes, accumulate with age. Their senescence-associated secretory phenotype (SASP) reshapes local microenvironments and can contribute to the systemic, low-grade inflammatory state often referred to as inflammaging. The consequence for bone is not only altered mass, but altered mechanosensory capacity and mechanoresponsiveness that likely contributes to fragility and impaired repair.
My lab sits at the intersection of skeletal aging biology and mechanics, with a deliberate focus on osteocytes because they are the longest-lived bone cells and the primary mechanosensors of mineralized tissue. My research is driven by a fundamental question in skeletal aging: when an osteocyte becomes senescent, what changes physically about its mechanosensation under load? To answer it, we pair molecular readouts with live single-cell biomechanical phenotyping using optical fiber interferometry nanoindentation on the Optics11 Life Pavone platform integrated on an inverted fluorescence microscope. This setup is central to our approach because it enables us to measure a cell’s mechanical phenotype while simultaneously capturing morphometric phenotype and fluorescence-based cell state identification in the same live cell.
Using this innovative approach, we found that stress-induced senescence in osteocytes is accompanied by a progressive biophysical shift, including cytoskeletal stiffening, altered plasma membrane viscoelastic behavior, and changes in adhesion forces (Figure 1a-d) [Aging Cell]. These changes are consistent with altered force transmission to mechanosensitive machinery and a shifted force-to-signal relationship in mechanotransduction.
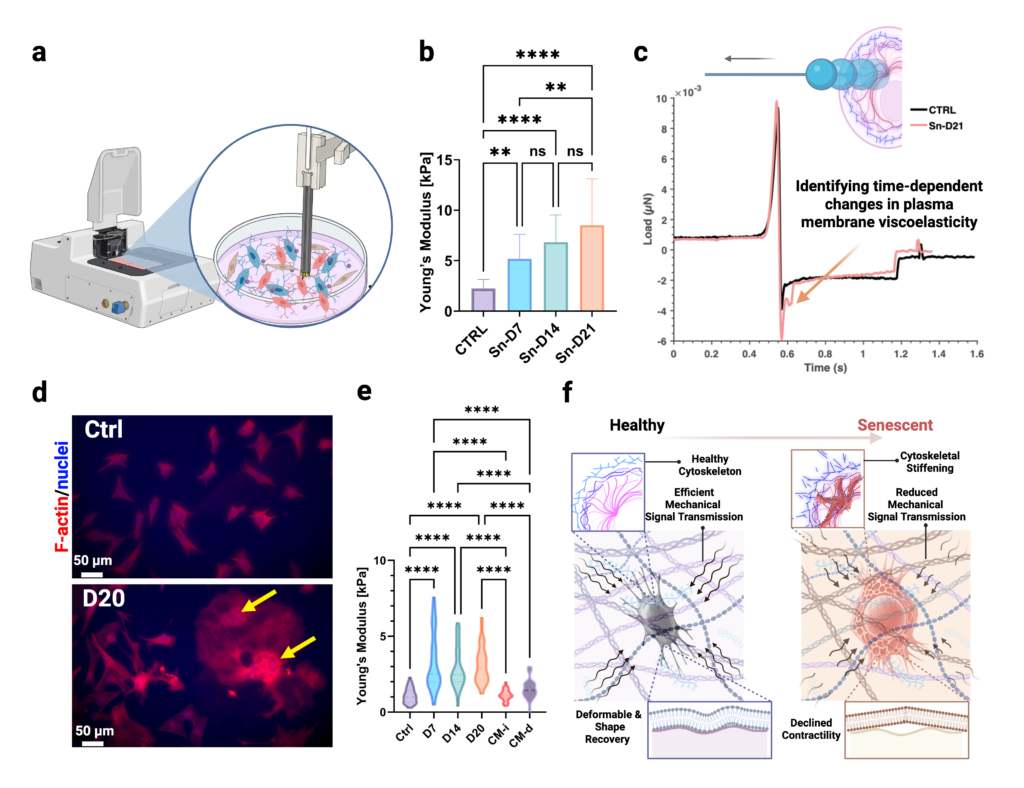
Figure 1 | Bone mechano-aging at single-cell resolution: senescence and SASP proximity drive distinct osteocyte mechanophenotypes. (a) Live single-cell optical fiber interferometry nanoindentation on the Optics11 Life Pavone platform enables time-lapsed biomechanical phenotyping in osteocytes. (b) Stress-induced senescence produces a time-dependent increase in osteocyte cytoskeletal Young’s modulus, consistent with progressive stiffening. (c) Representative unloading trace highlights senescence-associated changes in plasma membrane viscoelastic recovery and adhesion behavior. (d) Representative F-actin/nuclear immunostaining shows senescence-linked cytoskeletal remodeling and morphologic disruption at late time points. (e) Spatial-context experiment: transient paracrine exposure to SASP-enriched conditioned media alters inflammatory/matrix-remodeling expression signatures in recipient osteocytes [Small], yet single-cell stiffness remains near control; in contrast, persistent local accumulation of senescent factors results in pronounced stiffening and increased heterogeneity, consistent with a functional threshold for mechanical dysfunction. (f) Senescence-associated cytoskeletal stiffening and altered membrane mechanics reduce efficient mechanical signal transmission and contractile recovery, consistent with impaired mechanoresponsiveness [Aging Cell].
A second insight came from spatial context. We asked how SASP exposure affects otherwise non-senescent osteocytes. Transient paracrine exposure to senescent conditioned media altered inflammatory and matrix-remodeling gene expression profiles in recipient cells, but single-cell mechanical readouts were largely unchanged. In contrast, when senescent factors accumulated locally and persistently, recipient osteocytes developed significant biomechanical and structural deficits, including increased stiffness (Figure 1e), greater heterogeneity in cytoskeletal organization, and loss of dendritic integrity consistent with impaired mechanosensation [Small]. These findings indicate that a molecular SASP response is not automatically equivalent to mechanical dysfunction. Proximity and persistence of SASP exposure appear to be key determinants of when recipient osteocytes cross a functional threshold.
This is the lens we are building toward, which I call ”skeletal mechano-aging”. Molecular markers report which pathways are active. Single-cell biomechanical phenotypes report functional capacity, including how force is sensed, transmitted, dissipated, and converted into signaling. That distinction matters for understanding chronic disease progression as well as for designing repair strategies. Bone mineral density captures quantity, but it does not report the mechanosensory state of osteocytes or the local mechanical constraints that shape their response.
The same logic guides our regenerative work. In large or poorly vascularized defects, early healing unfolds under two coupled constraints: oxygen limitation and a mechanically evolving provisional matrix. In our recent Advanced Healthcare Materials study, we developed a visible-light DLP 3D-printed hydrogel scaffold with a triply periodic minimal surface (TPMS) architecture, embedded with CaO2-loaded hollow silica nanoparticles to provide localized, short-term oxygen release during the early hypoxic phase. The TPMS architecture governs transport and deformation, while the silica-shelled, oxygen-releasing nanoparticles provide localized oxygenation and mechanically reinforce the hydrogel. Using the Pavone platform, we tracked micromechanical evolution as the scaffold remodeled and linked those measurements to in vivo healing readouts in a murine calvarial defect model, where the system improved repair without detectable systemic toxicity (Figure 2).
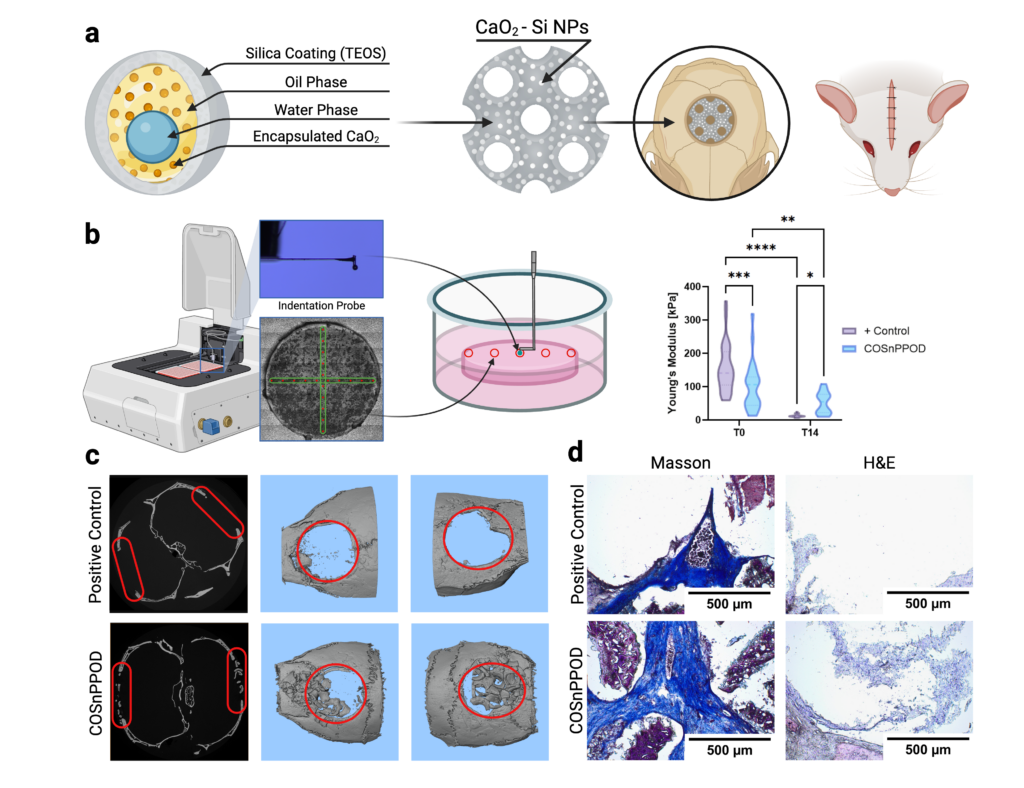
Figure 2 | Engineered repair microenvironment: oxygen-releasing, mechanically reinforced TPMS hydrogel improves defect healing. (a) Core-shell CaO₂-loaded, silica-coated nanoparticles designed for controlled oxygen release and mechanical reinforcement, embedded within a visible-light DLP 3D printed TPMS hydrogel scaffold for calvarial defect repair. (b) Pavone-enabled micromechanical mapping of the scaffold in vitro, showing Young’s modulus evolution over time (T0 vs T14) and highlighting nanoparticle-dependent stiffness tuning during remodeling/degradation. (c) Representative μCT reconstructions and defect-region quantification demonstrating enhanced bone repair in the oxygen-releasing TPMS scaffold relative to controls. (d) Histology (Masson’s trichrome and H&E) supporting improved tissue deposition and bridging in the oxygen-releasing TPMS scaffold group [Advanced Healthcare Materials].
Across these projects, we are pursuing one question at two scales: how the aging skeleton converts load into biological signals, and how that mechanobiology should shape strategies for repair. Our long-term direction is to use single-cell mechanical signatures, paired with morphometric and molecular phenotypes, as design inputs for both in vitro aging models that isolate defined cell states and minimally invasive and 3D bioprinted repair systems tuned for senescent-rich tissue. The goal is not only regenerated bone, but restored, load-responsive remodeling that remains durable with age.

Maryam Tilton, PhD, is an Assistant Professor in the Walker Department of Mechanical Engineering at The University of Texas at Austin and Director of the BioMATTER Lab. She is a member of the Lawrence Family Bone Disease Program of Texas and completed an NIH-T32 postdoctoral fellowship in musculoskeletal health at Mayo Clinic. Her lab studies how aging and senescence alter osteocyte-matrix mechanobiology and leverages those mechanistic insights to engineer microenvironments and minimally invasive strategies that improve skeletal repair and reduce fracture risk in aging adults. She is a recipient of an National Institute on Aging (NIA) Research Career Development Award.
maryamtilton.com
Interested in presenting your research in Faces of Mechanobiology?
Send an email to marketing@optics11life.com
Mentioned in this article:
Pavone
Pavone is automated and optimized for the biological workflow. It is a high-throughput, accurate, and easy-to-use solution that measures the local mechanical properties of cells, biomaterials, and 3D in vitro models.

