Brain organoids are transforming neuroscience and disease research, and even opening new directions in biohybrid computing. Yet many of the biological and information-processing behaviors that make organoids so compelling are happening in physical tissue that deforms, resists load, and interacts mechanically with its surroundings. When organoids are grown in hydrogels, integrated into microphysiological devices, fused into assembloids, or penetrated by electrodes, their mechanical properties become part of the experiment.
In this Faces of Mechanobiology entry, John Finan, PhD shares why organoid biomechanics is a missing piece of the puzzle, and how their lab took a first step toward quantifying it. Using spherical indentation with the Optics11 Life Chiaro nanoindenter, they move from an initial elastic characterization to viscoelastic modeling suited to rate-dependent behavior, and report how stiffness and mechanical response vary across organoid batches and cell lines. The result is a foundation for more biofidelic mechanical environments, and for understanding how deformation and stress may shape organoid biology in development, disease, and traumatic brain injury models [REF 1, REF 2].
Beyond “Squishy Balls”: Quantifying Brain Organoid Biomechanics
Brain organoids have an exciting capacity to reproduce the function and development of the human brain in vitro. This capacity has, unsurprisingly, attracted the attention of neuroscientists and developmental biologists who have quickly created a vast body of knowledge about how organoids form circuits and how cells mature and differentiate within them. This knowledge has already been used to advance our understanding of important neurological diseases ranging from epilepsy to bipolar disorder to neurodegenerative conditions. Organoids also have the capacity to function as reservoir computers: devices that can accept low-dimensional inputs and map them on to high-dimensional outputs. This mapping dramatically accelerates the training of artificial neural networks so it can enable more energy-efficient computing. Significantly, the complex biological and information handling processes that occur in organoids do not happen in an abstract space. They happen in little squishy balls of brain cells. Our current knowledge of the biomechanics of organoids does not go much beyond this vague description.
The mechanical properties of organoids matter whenever they touch something else. When organoids are placed in a microphysiological device or a hydrogel environment, the extent to which they deform the environment, or the environment deforms them depends on their mechanical properties. Electrodes that pierce organoids should be as thin as possible to minimize damage, but not so thin that they buckle before penetration. Optimizing this trade-off requires organoid mechanical properties. Organoids are also frequently grown in contact with other organoids. Such a culture is called an assembloid. For example, an organoid model of the brain might be grown in contact with an organoid model of the spinal cord so that they spontaneously fuse and reproduce interactions between the brain and spinal cord during development. Many cell types are durotactic, i.e. they migrate along stiffness gradients. Therefore, cell migration within an assembloid might depend on the relative stiffnesses of its component organoids. Organoids have also been transplanted into rodent brains. A key advantage of this procedure is that rodent blood vessels infiltrate the human organoid tissue and nourish it. Angiogenesis is also a mechanically sensitive process. Our lab uses organoids to model traumatic brain injury, so we are particularly motivated to understand how stresses in the tissue increase as the extent and rate of tissue deformation increases. Given the paucity of data in the field, we decided the first step was quantification of the mechanical properties of the tissue.
We started with the assumption that brain organoids would be qualitatively similar to primary brain tissue, which we have studied in the past. Brain tissue is a challenging material to characterize because its properties are rate-dependent, anisotropic, and non-linear and its microstructure is complex. Our approach was to start with the simplest possible model and then advance incrementally towards more complex models as dictated by scientific necessity. We began with a spherical indentation procedure using the Optics11 Life Chiaro nanoindenter and an initial elastic characterization using the Hertz contact solution.
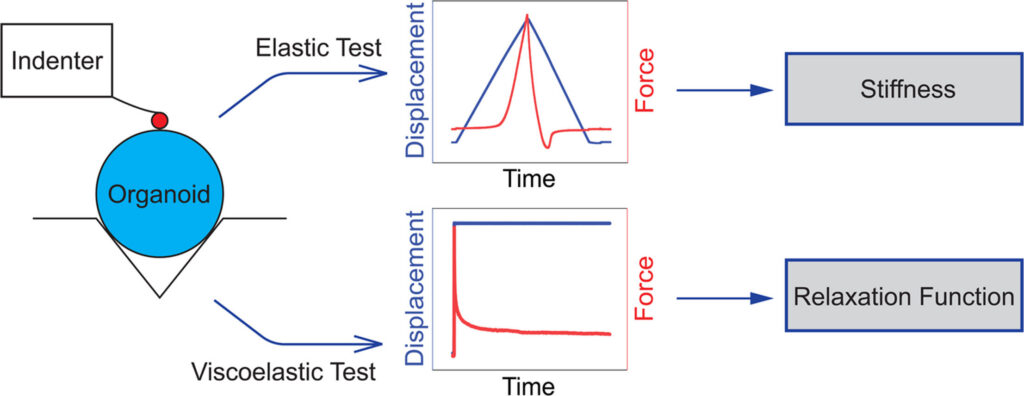
Figure 1 summarizes the indentation workflow, showing the elastic test used to estimate stiffness and the ramp-and-hold test used to extract the viscoelastic relaxation function.
From there, we proceeded to a viscoelastic model because rate-dependent effects are important in our study of traumatic brain injury phenomena. To measure these properties, we applied a ramp-and-hold indentation protocol and fit the declining force history to a quasi-linear viscoelastic version of the Hertz contact solution. Our results showed that brain organoids have mechanical properties on the low end of the wide range of properties that have been reported for primary brain tissue. This conclusion is unsurprising since immature brain tissue is typically more compliant than mature brain tissue and brain organoids mimic the fetal stage of brain development. The tissue was highly rate-dependent, as is primary brain tissue, and properties varied between batches of organoids derived from the same cell line and between cell lines, indicating that these influences should not be ignored in biomechanical studies.
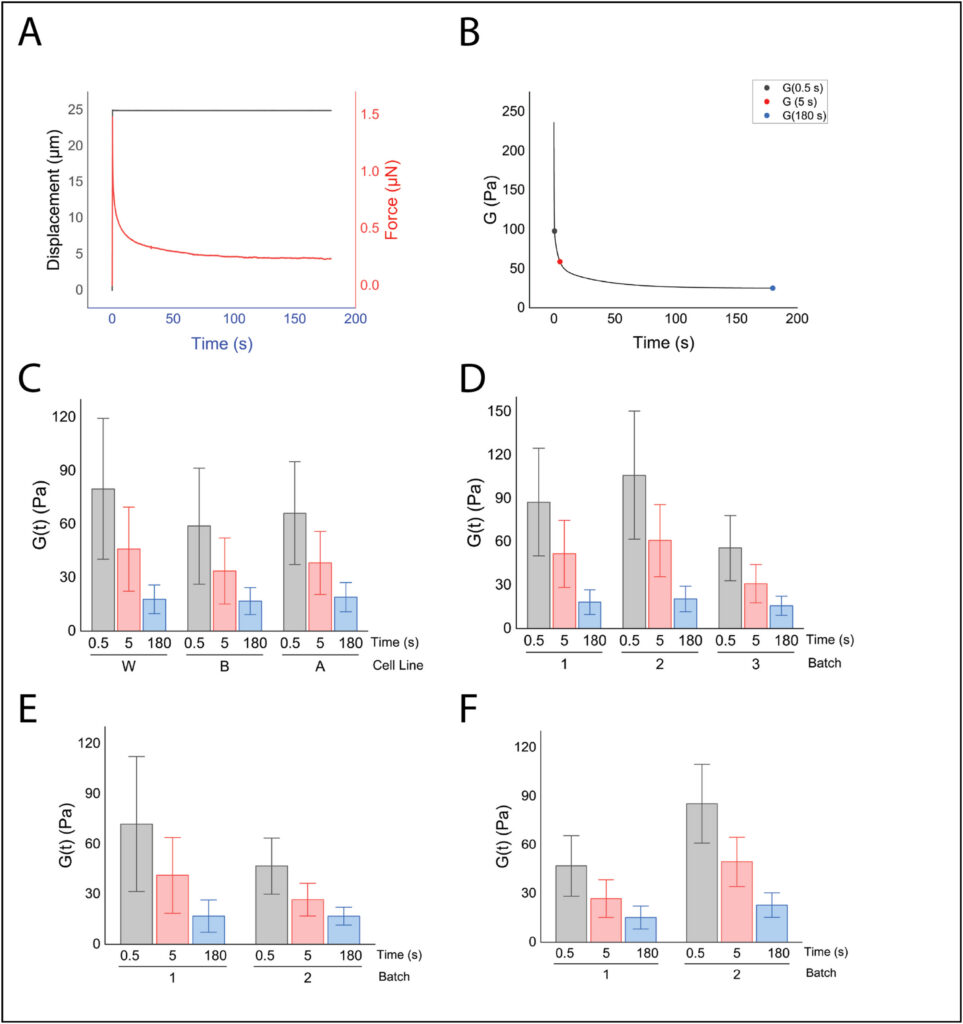
Figure 2 shows the ramp-and-hold response and derived relaxation modulus, and highlights how properties shift across time scales as well as between cell lines and batches.
These results provide a starting point for future mechanical investigations, but many important questions remain about how organoids deform under load, and how those deformations influence their biology. Great care and effort has been invested in optimizing the chemical signals in the organoid environment so that the concentration of growth factors and other cues matches the in situ condition. However, the mechanical environment is typically less biofidelic, consisting of a fluid medium with an effective stiffness of zero and none of the pulsatile loads applied by the vasculature in situ. The influence of the biomechanical environment on the biological and information handling processes in organoids that have generated so much excitement about organoids remains largely unexplored.
References
- Shiravi S, Yufa A, Papavasileiou P, Lotz S, Murphy D, Bertucci T, Temple S, Finan JD. A Model of Traumatic Brain Injury Oligomerizes Tau in Cortical Organoids and Induces Clinically Relevant Pathologies that Synergize with MAPT Mutation. J Neurotrauma. 2025 Nov;42(21-22):2015-2029. doi: 10.1177/08977151251374286. Epub 2025 Sep 6. PMID: 40900146; PMCID: PMC12836475.
- Shoemaker AR, Jones IE, Jeffris KD, Gabrielli G, Togliatti AG, Pichika R, Martin E, Kiskinis E, Franz CK, Finan JD. Biofidelic dynamic compression of human cortical spheroids reproduces neurotrauma phenotypes. Dis Model Mech. 2021 Dec 1;14(12):dmm048916. doi: 10.1242/dmm.048916. Epub 2021 Dec 22. PMID: 34746950; PMCID: PMC8713991.

John D. Finan, PhD.
John Finan is an Assistant Professor in the Department of Mechanical and Industrial Engineering at the University of Illinois Chicago. He studies the biomechanics of the human brain, particularly as it relates to understanding and treating traumatic brain injuries and their long-term consequences. He uses human brain cells and organoids (self-organizing aggregates of thousands of brain cells) derived from stem cells in his research because they allow him to answer questions about how human brains differ from animal brains, and why one human’s brain differs from another’s. He received his PhD. from Duke University, where he published on improving motorcycle helmets and the biomechanics of cartilage. He also held a post-doctoral appointment at Columbia University in New York, where he studied the biomechanics of the rat, pig, and human brains and worked on a novel approach to treating brain swelling after trauma. His work has been funded by the National Institutes of Health and the National Science Foundation.
Interested in presenting your research in Faces of Mechanobiology?
Send an email to marketing@optics11life.com
Mentioned in this article:
Chiaro
Chiaro is a microscope-compatible, nanoindentation system that allows you to combine unique mechanical insights with the imaging equipment of your choice. A compact yet powerful instrument to expand your lab infrastructure.

