Living tissues continuously sense, generate, and respond to force. In health, this ongoing dialogue between cells and their physical environment can settle into mechanical homeostasis, a balanced state where forces, material properties, and cellular responses remain stable over time. When that balance slowly drifts, tissues may still look structurally intact, yet operate closer to a failure threshold, increasing vulnerability to ageing-related decline and disease.
In this Faces of Mechanobiology entry, Jacopo Di Russo shares how the retina offers a powerful model system to study mechanical homeostasis. He explains why retinal mechanics cannot be reduced to stiffness alone, and how cells integrate gradients, anisotropy, time-dependent deformation, and coupled biochemical and mechanical cues. Drawing on recent work in the outer retina, including regional mechanical differences in the retinal pigment epithelium (RPE) and in vitro models of ageing-related extracellular matrix deformation and reduced RPE cell density, Jacopo shows how mechanobiology can reveal early vulnerability and potential intervention points.
He also highlights how integrating quantitative mechanical measurements into standard workflows, using tools such as the Optics11 Life Chiaro mounted on an epifluorescence microscope, can help turn mechanical homeostasis from an abstract concept into a measurable biological variable.
Mechanical Homeostasis: Lessons from the Retina
Living tissues are not static structures. They are dynamic mechanical systems that constantly sense, generate, and respond to forces. Under healthy conditions, this dialogue between cells and their physical environment settles into what we often call mechanical homeostasis: a state in which forces, material properties, and cellular responses remain balanced over time.
What fascinates me is not only how tissues reach and maintain this equilibrium, but what happens when they slowly drift away – a process that can actively drive ageing and disease.
Mechanical homeostasis emerges from the interplay between cells and their surroundings. Cells pull on the extracellular matrix, remodel it, stiffen or soften it, and respond to its biochemical and physical cues through mechanotransduction pathways. At the same time, cell–cell junctions distribute forces across tissues, enabling collective responses. This balance is actively regulated: cells adapt their cytoskeleton, adhesion, and contractility to keep mechanical stresses within a tolerable range. When successful, tissues remain functional and resilient; when it fails, they may enter a new mechanical regime that appears stable but is biologically maladaptive.
My team focuses on the retina, a tissue that provides a particularly powerful model to study mechanical homeostasis. It is a highly organized, layered tissue with extreme functional demands and minimal regenerative capacity. From a mechanical perspective, the retina sits at the interface between soft neural tissue, supportive extracellular matrices, and fluid environments, requiring continuous adaptation to tension, pressure, and matrix properties while preserving precise organisation.
The retina is postmitotic: its cells cannot re-enter the cell cycle. Over time, natural cell death gradually reduces cell number, necessitating tissue restructuring that challenges mechanical equilibrium. This makes the retina an ideal system for asking: how do tissues maintain mechanical balance over decades, and what happens when this regulation slowly fails? Unlike many other tissues, small structural or mechanical changes in the retina can have disproportionately large functional consequences.
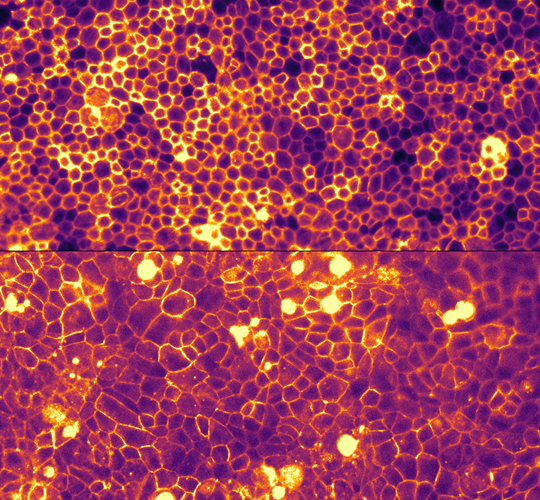
Figure 1: The retinal pigment epithelium (RPE), located adjacent to the light-sensitive photoreceptors, can be modelled in vitro using stem cell–derived RPE cells. In culture, these cells become postmitotic, allowing us to study mechanical homeostasis in the tissue. The top panel shows young RPE, while the bottom panel illustrates reduced cell density, mimicking an aged state.
When homeostasis drifts: ageing and disease risk
Ageing is often discussed in molecular or genetic terms, but it also has a mechanical dimension. Over time, extracellular matrices stiffen, crosslinking increases, and tissue viscoelasticity changes. Cells, however, do not always adapt proportionally. Such mechanical drift can affect how cells sense their environment, how forces are transmitted, and how mechanosensitive pathways are engaged. These changes may occur long before overt pathology becomes visible.
A tissue that has shifted mechanically may appear structurally intact yet operate closer to a failure threshold. Additional stress, such as metabolic, inflammatory, or genetic, can then tip the system into degeneration. Rather than viewing disease as sudden breakdown, this perspective frames it as the endpoint of long-term imbalance in mechanical regulation.
Mechanics is not just about stiffness. Cells respond to gradients, anisotropies, time-dependent deformations, and the coupling between biochemical and mechanical signals. Mechanical homeostasis, therefore, cannot be reduced to a single parameter. Understanding how cells integrate these cues, and how this changes with age, is essential if we want to intervene meaningfully.
Our work reveals a key role for mechanics in the outer retina. We demonstrated that the mechanical state of the retinal pigment epithelium (RPE) differs across visual angles, in agreement with regional variations in extracellular matrix composition and cellular workload. These adhesion-dependent properties regulate the capacity of RPE cells to apically support photoreceptor cells (Kozyrina A. and Piskova T. et al. EMBO reports 2025). More recently, we modelled in vitro ageing-related phenomena of the outer retina, including local topographical deformations of the extracellular matrix (Reul F. et al. Small 2026) and the gradual reduction in RPE cell density (Piskova T. et al. Nature Communications 2026). Each of these processes independently challenges tissue mechanical homeostasis, ultimately resulting in functional decline.
Taken together, our findings demonstrate how mechanobiology offers a powerful framework for rethinking ageing and disease. By focusing on how tissues maintain equilibrium, rather than only what breaks, we gain new insight into early vulnerability, resilience, and potential intervention points.
Advances in experimental tools and models for probing mechanics at cellular and tissue scales made this work possible. Technologies developed by companies such as Optics11 Life allowed us to integrate quantitative mechanical measurements into standard workflows. Using a Chiaro instrument mounted on an epifluorescence microscope, we performed precise mechanical characterisation of stem cell–derived tissues and ECM-mimicking hydrogels alongside conventional imaging. This integration enabled mechanical homeostasis to be treated not as an abstract concept, but as a measurable biological variable that may ultimately help us predict — and perhaps even prevent — disease risk before irreversible damage occurs.
References
Kozyrina A. and Piskova T. et al. EMBO reports: https://doi.org/10.1038/s44319-025-00475-9
Reul F. et al. Small 2026: https://doi.org/10.1002/smll.202509246
Piskova T. et al. Nature Communications 2026: https://doi.org/10.1038/s41467-026-71493-x
PD. Dr. Jacopo Di Russo
Jacopo Di Russo studied Cell and Molecular Biology in Florence, Italy, before joining Prof. Lydia Sorokin’s lab in Münster, Germany, as a Marie Curie Fellow, where he explored how the extracellular matrix shapes physiological responses. In 2015, he moved to Prof. Joachim Spatz’s lab at the Max Planck Institute for Medical Research in Heidelberg to deepen his expertise in biophysical cell adhesion. In 2019, he founded the Retinal Mechanobiology and Disease group (REMeD), supported by a competitive grant from the RWTH Aachen Medical Faculty. He is also associated with the DWI – Leibniz Institute for Interactive Materials, bridging material science and chemical engineering with medical biology to tackle questions in retinal physiology.
For more information, visit www.dirussolab.org
Interested in presenting your research in Faces of Mechanobiology?
Send an email to marketing@optics11life.com
Mentioned in this article:
Chiaro
Chiaro is a microscope-compatible, nanoindentation system that allows you to combine unique mechanical insights with the imaging equipment of your choice. A compact yet powerful instrument to expand your lab infrastructure.


